Antiviral effect of "npi Antivirus Paper" on the novel coronavirus
Nippon Paper Industries Co., Ltd.
Nippon Paper Industries Co., Ltd. (head office : Chiyoda-ku, Tokyo,President and Representative Director: Toru Nozawa, hereinafter referred to as"the Company") announced that "NPI AntiviralPaper"NPI AntiviralPaper". It has been confirmed that it has an antiviral effect against the new coronavirus.
■Effects on the new coronavirus SARS-CoV-2 (new coronavirus infection COVID19)
A team led by Assistant Professor Ryohei Hirose at Kyoto Prefectural University of Medicine evaluated the survival time of the novel coronavirus on the Paper surface of NPI antiviral Paperand found that the survival time of the virus was significantly shortened on the Paper surface of NPI antiviral Paper compared to ordinary high-qualityPaper.
- Virus stock solution SARS-CoV-2
・Adjust the viral solution 5.0×107 (TCID50/ml) 1 μL, PBS (phosphate-buffered saline) 19 μL
・Virus application amount: Adjust the virus solution 2μL (1.0×105TCID50)
・Contact time: Collected in DMEM (Dalbecco Modified Eagle Medium) + 5% FCS (fetal bovine serum) 1000μL at any elapsed time.
Virus survival time on each Paper sample surface
| Ping Volume (g/m2) |
General High QualityPaper 186.1 |
NPI AntiviralPaper |
|---|---|---|
| Viral survival time |
3624.3 points |
150.9 points |
Stability of the novel coronavirus on the surface of each Paper sample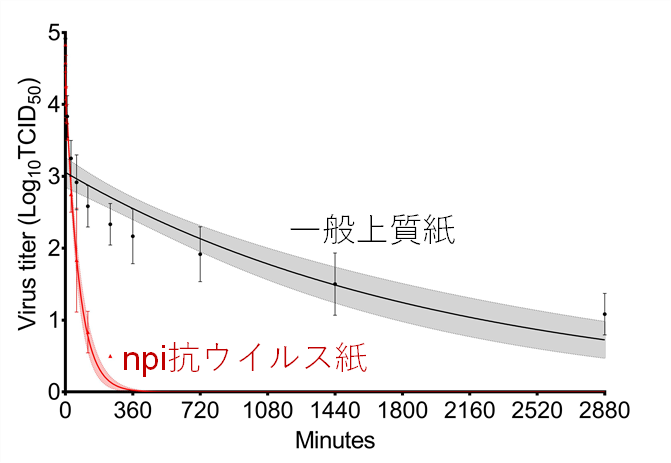
This product can be printed and processed in the same way as regularprinting paper, so it can provide safety and security in a variety of applications. It was launched in September 2020 and obtained SIAA antiviral processing certification in December 2021. It can be used for placemats, paper files, notebooks, mask cases, business cards, etc.
■Lineup
Brand Name ; NPI AntiviralPaper
Handling Ping Capacity ; 80g/㎡、186.1g/㎡
■Related Links
npi AntiviralPaperReceives SIAA Antiviral Processing Certification (December 17, 2021)
"Antiviral" printing paper "NPI AntiviralPaper" with performance was developed (dated September 10, 2020)
